|
Celebrating 33 years providing high quality products and advice.
|
| Our Local Time Is 9:35:07 AM. |
| Call us at 818-786-0600. We are here to help! |
 |
ON SALE NOW
Introducing the Polaris Lab Water Systems
High Purity Water Made In The USA.
Click here for more info. |
Heavy Water
|
Even though its molecular formula suggests that water is a simple compound it is in fact not that simple. Water is actually a mix of hydrogen and oxygen isotopes that create a waterlike mixture. Hydrogen an oxygen both have both have three isotopic forms. This means that water consists of 18 different molecular combinations.
Present in minute quantities the molecular for composed of hydrogen has an atomic weight of 1 while oxygen has an atomic weight of 16.
However with atomic energy one of the isotopic forms of water became very important. In 1932 Harold Urey discovered the isotopic molecule that consisted of an oxygen atom with an atomic weight of 16 and two hydrogen atoms with an atomic weight of 2. He named it 'heavy Water.' He discovered that the nuclei of the hydrogen atoms contained a neutron in addition to a proton.
Due to its slightly different properties than a typical hydrogen atom, this isotope was named deuterium.
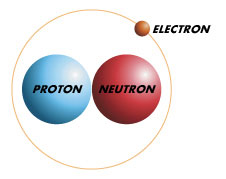
Heavy water has different properties that distinguish it from ordinary water. To begin with it boils at 101.4oC and freezes at 3.8oC.It is physically inert thus it will not support the life processes. however its most important property is that it has an ability to functoin as a moderator in atomic fission.
For example with fission and uranium 235 slow neutrons are much more effective for splitting heavy nuclei than faster neutrons. In order to have a and sustain a chain reaction that is initiated by fast neutrons that are given off by uranium 235 in its initial fission a moderator is needed to slow down the neutrons without capturing them. Regular is not conducive to this because the hydrogen nuclei capture the faster neutrons thus slowing them down. The ideal moderator in this is instance is heavy water due to the fact that is absorbs large portions of the neutrons energy without capturing the neutron for itself as in ordinary water. Produced by a catalytic exchange between hydrogen and gas, heavy water plays a major role in all of the atomic processes.
More Information About Water
Molecular Structure
The Three States
Sublimation
Supercooled Water
Heavy Water
Properties
Heat Capacity
Latent Heats
Solvent
Surface Tension
| |
|
Images are representative of the products. Images may or may not be of the actual product. If it is important e-mail us for an actual image if available.
* Flat Rate UPS shipping when able to ship via UPS and is in the USA excluding Hawaii and Alaska.
Larger Items may not be able to ship via UPS, in that case freight charges will be quoted seperately.
International shipping will be quoted after the order is placed. You will have the opportunity to cancel before we finalize your order.
Terms and conditions
Credit Application
Privacy
Policy
List All Products
|













